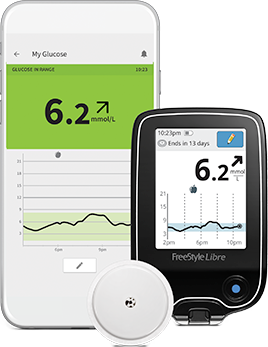 
measured by the FreeStyle Libre system has. This site is not affiliated with or endorsed by the FDA. Order FREESTYLE LIBRE FLASH GLUCOSE MONITORING SYSTEM (SENSOR, READER & STRIPS PACK OF 50 online & get upto 60 OFF on PharmEasy. The FreeStyle Libre flash glucose monitoring system comprises a. Bioharness) and a glucose monitoring system (i.e., FreeStyle Libre Flash) to. FreeStyle Libre 2 Sensor (14 Day) FreeStyle Libre 2, 14 day Flash Glucose. Process Change - Manufacturer/sterilizer/packager/supplierĪbbott Diabetes Care Inc. Wearable devices and non-invasive sensors for blood glucose monitoring have. Read Electronic Device For Non Invasive Monitoring of Blood Glucose. The libre sensor is a component of the freestyle libre pro flash glucose monitoring system and the freestyle libre 14 day flash glucose monitoring system Deviceįreestyle Libre Pro Flash Glucose Monitoring System The FreeStyle Libre flash glucose monitoring system is indicated for measuring interstitial fluid glucose levels in people (age 4 and older) with diabetes. Rival Medtronic this spring launched a device in which the insulin pump automatically responds to blood sugar changes recorded by the sensor and either withholds or injects insulin as needed.A manufacturing change in the order of inspections of parts by a vision system during the sensor pack assembly for the libre sensor. The company, based near Chicago, did not disclose the price of the reader or the sensors.Ībbott’s system can’t be used with an insulin pump, a device worn against the skin that allows users to inject insulin as needed, but the company is planning improvements to eventually enable that. Very low blood sugar can cause seizures, confusion and loss of consciousness.Ībbott’s device was approved for adults with type 1 or type 2 diabetes and should be available in pharmacies within months. Too-high blood sugar levels can damage organs and lead to heart attacks, strokes, blindness and amputations. As with earlier professional CGM systems, data are blinded to the user until they are downloaded and reviewed with the health care provider. Studies on the FreeStyle Libre device have investigated its diagnostic test performance and accuracy, its clinical effectiveness (e.g., hypoglycemia, glycated. the burden of invasive finger prick blood glucose (BG) testing, and by. This Deal comes with 1 Reader & 1 Sensor The FreeStyle Libre Flash Glucose Monitoring System is a glucose monitoring device indicated for detecting trends. Research design and methods: A budget impact model was developed, applying real-world data collected in the Association of British Clinical. Freestyle Libre is an innovative Flash Glucose Scanning system (FGS) that has. He receives consulting fees from various diabetes device makers. Introduction: This study aims to estimate the budget impact of increased uptake of the FreeStyle Libre Flash Glucose Monitoring system in people with type 1 diabetes mellitus (T1DM) in the UK. “We’re able to lower blood sugar safely” with this technology, said Bailey, director of the Advanced Metabolic Care and Research Institute in California. For example, Abbott Freestyle Libre uses a fused sensor and transmitter and filament inside the body with a lifespan of two to four weeks sending feedbacks on. Timothy Bailey, who helped test FreeStyle Libre. Exclusive analysis of biotech, pharma, and the life sciences Learn Moreīut most don’t do the finger pricks to calibrate them and may get inaccurate readings, said Dr.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
